Lagoon Systems - 4...
Lagoon Microbiology...
Microorganisms found in aerated and facultative ponds are more diverse than those observed in many other biological
treatment processes, due to the diverse growth environments present. Both aerobic and anaerobic bacteria are involved
as well as algae and some higher life forms such as protozoans, rotifers, daphnia, and insect larvae.
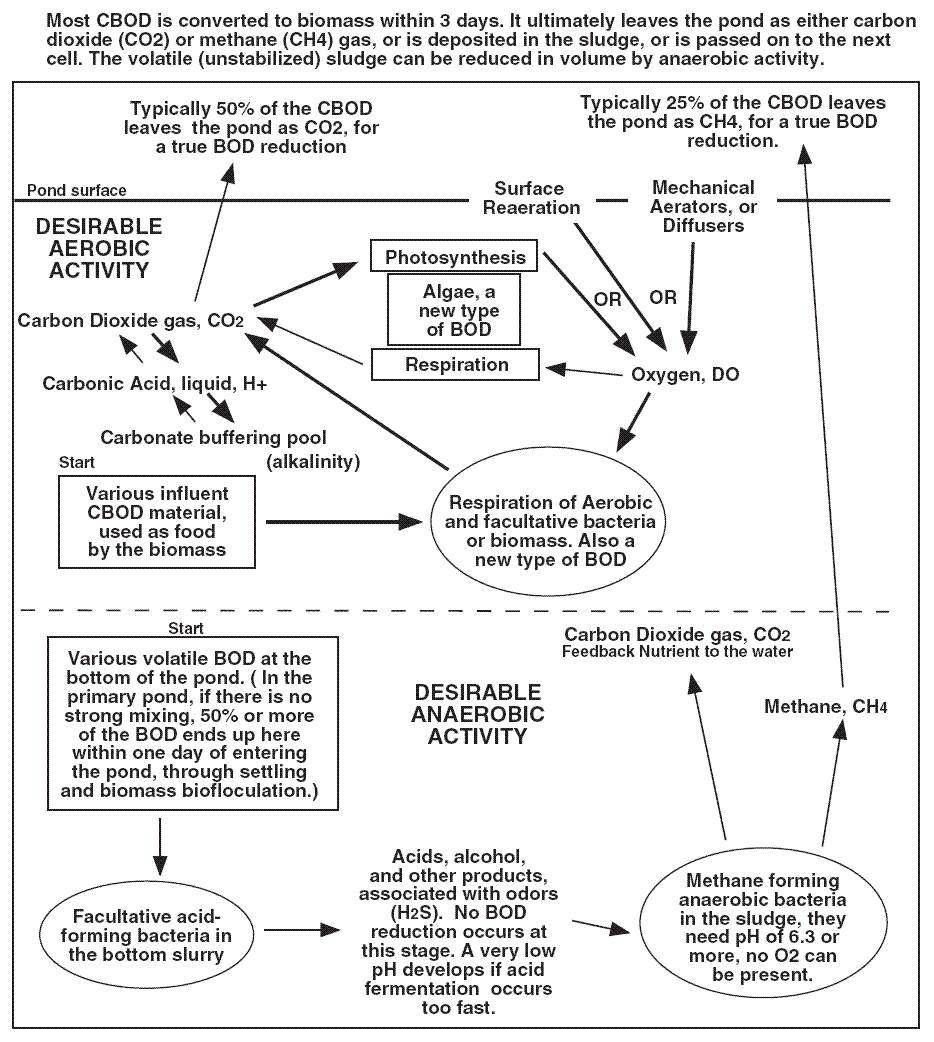 Aerobic Bacteria...
The aerobic bacteria that occur are similar to those found in other treatment processes such as activated sludge.
Three functional groups occur : freely dispersed, single bacteria; floc-forming bacteria; and filamentous bacteria.
All function similarly to oxidize organic carbon (BOD) to produce CO 2 and new bacteria (new sludge).
Aerobic Bacteria...
The aerobic bacteria that occur are similar to those found in other treatment processes such as activated sludge.
Three functional groups occur : freely dispersed, single bacteria; floc-forming bacteria; and filamentous bacteria.
All function similarly to oxidize organic carbon (BOD) to produce CO 2 and new bacteria (new sludge).
Many bacterial species that degrade wastes grow as single bacteria dispersed in the wastewater. Although these readily oxidize BOD, they do not settle and hence often leave the lagoon system in the effluent as solids (TSS). These tend to
grow in lagoons at high organic loading and low oxygen conditions. More important are the floc-forming bacteria, those
that grow in a large aggregate (floc) due to exocellular polymer production (the glycocalyx). This growth form is
important as these flocs degrade BOD and settle at the end of the process, producing a low TSS effluent.
A number of fiIlamentous bacteria occur in lagoons, usually at specific growth environments. These generally do not cause
any operational problems in lagoons, in contrast to activated sludge where filamentous bulking and poor sludge settling
is a common problem.
Most heterotrophic bacteria have a wide range in environmental tolerance and can function effectively in BOD
removal over a wide range in pH and temperature. Aerobic BOD removal generally proceeds well from pH 6.5 to 9.0
and at temperatures from 3 - 4 O C to 60 - 70 O C (mesophilic bacteria are replaced by
thermophilic bacteria at temperatures above 35 O C). BOD removal generally declines rapidly below
3 - 4 O C and ceases at 1 - 2 O C. A very specialized group of bacteria occurs to some extent
in lagoons (and other wastewater treatment systems) that can oxidize ammonia via nitrite to nitrate, termed nitrifying bacteria. These bacteria are strict aerobes and require a redox potential of at least + 200 m V (Holt et al., 1994).
It was once thought that only two bacteria were involved in nitrification : Nitrosomonas europaea, which oxidizes
ammonia to nitrite, and Nitrobacter winogradskyi, which oxidizes nitrite to nitrate. It is now known that at least
5 genera of bacteria oxidize ammonia and at least three genera of bacteria oxidize nitrite (Holt et al., 1994).
Besides oxygen, these nitrifying bacteria require a neutral pH ( 7 - 8 ) and substantial alkalinity ( these autotrophs
use CO 2 as a carbon source for growth). This indicates that complete nitrification would be expected at pond
pH values between pH 7.0 and 8.5. Nitrification ceases at pH values above pH 9 and declines markedly at pH values below 7. This results from the growth inhibition of the nitrifying bacteria. Nitrification, however, is not a major pathway for nitrogen removal in lagoons. Nitrifying bacteria exists in low numbers in lagoons. They prefer attached growth systems
and / or high MLSS sludge systems.
Anaerobic Bacteria...
Anaerobic, heterotrophic bacteria that commonly occur in lagoons are involved in methane formation (acid-fonning and
methane bacteria) and in sulfate reduction (sulfate reducing bacteria). Anaerobic methane formation involves three
different groups of anaerobic bacteria that function together to convert organic materials to methane via a three step process. General anaerobic degraders - many genera of anaerobic bacteria hydrolyze proteins, fats, and poly saccharides present in wastewater to amino acids, short-chain peptides, fatty acids, glycerol, and mono- and di-saccharides. These
have a wide environmental tolerance in pH and temperature. Acid-forming bacteria - this diverse group of bacteria converts products from above under anaerobic conditions to simple alcohols and organic acids such as acetic, propionic, and butyric. These bacteria are hardy and occur over a wide pH and temperature range. Methane forming bacteria - these bacteria convert formic acid, methanol, methylamine, and acetic acid under anaerobic conditions to methane. Methane is derived in part from these compounds and in part from CO 2 reduction. Methane bacteria are environmentally sensitive and have a
narrow pH range of 6.5 - 7.5 and require temperatures > 14 O C.
Note that the products of the acid formers (principally acetic acid) become the substrate for the methane producers.
A problem at times exists where the acid formers overproduce organic acids, lowering the pH below where the methane
bacteria can function ( a pH < 6.5 ). This can stop methane fonnation and lead to a buildup of sludge in a lagoon with
a low pH. In an anaerobic fennenter, this is called a "stuck digester". Also, methane fennentation ceases at cold temperature, probably not occurring in most lagoons in the wintertime in cold climates. A number of anaerobic bacteria
(14 genera reported to date (Bolt et al., 1994)) called sulfate reducing bacteria can use sulfate as an electron acceptor, reducing sulfate to hydrogen sulfide. This occurs when BOD and sulfate are present and oxygen is absent. Sulfate reduction
is a major cause of odors in ponds.
Photosynthetic Organisms...
Anaerobic, photosynthetic bacteria occur in all lagoons and are the predominant photosynthetic organisms in anaerobic lagoons, The anaerobic sulfur bacteria, generally grouped into the red and green sulfur bacteria and represented by
about 28 genera (Ehrlich, 1990), oxidize reduced sulfur compounds ( H 2 S ) using light energy to produce
sulfur and sulfate, Here, H 2 S is used in place of H 2 O as used by algae and green plants,
producing S0 4 - instead of O 2 .
All are either strict anaerobes or microaerophilic. Most common are Chromatium, Thiocystis, and Thiopedia, which can
grow in profusion and give a lagoon a pink or red color. Finding them is most often an indication of organic overloading
and anaerobic conditions in an intended aerobic system. Conversion of odorous sulfides to sulfur and sulfate by these
sulfur bacteria is a significant odor control mechanism in facultative and anaerobic lagoons, and can be desirable.
Algae are aerobic organisms that are photosynthetic and grow with simple inorganic compounds
( CO 2 , NH 3 , NO 3 - , and PO 4 - 2 ) using
light as an energy source. Note that algae produce oxygen during the daylight hours and consume oxygen at night.
Algae are desirable in lagoons as they generate oxygen needed by bacteria for waste stabilization. Three major groups
occur in lagoons, based on their chlorophyll type: brown algae ( diatoms ), green algae, and red algae. The predominant
algal species at any given time is dependent on growth conditions, particularly temperature, organic loading, oxygen
status, nutrient availability , and predation pressures. A fourth type of "algae" common in lagoons is the cyano-bacteria
or blue-green bacteria. These organisms grow much as the true algae, with the exception that most species can fix
atmospheric nitrogen. Blue-green bacteria often bloom in lagoons and some species produce odorous and toxic by-products. Blue-green bacteria appear to be favored by poor growth conditions including high temperature, low light, low nutrient availability ( many fix nitrogen ) and high predation pressure. Common blue-green bacteria in waste treatment systems
include Aphanothece, Microcystis, Oscillatoria and Anabaena. Algae can bloom in lagoons at any time of the year ( even
under the ice) ; however, a succession of algal types occurs over the season.
There is also a shift in the algal species present in a lagoon through the season, caused by temperature and rotiter
and Daphnia predation. Diatoms usually predominate in the wintertime at temperatures < 60 O F. In the early
spring when predation is low and lagoon temperatures increase above 60 O F, green algae such as Chlorella, Chlamydomonas, and Euglena often predominate in waste treatment lagoons. The predominant green algae change to species
with spikes or horns such as Scenesdesmus, Micractinium, and Ankistrodesmus later in the season when Rotifers and
Daphnia are active ( these species survive predation better ).
Algae grow at warmer temperature, longer detention time, and when inorganic minerals needed for growth are in excess. Alkalinity ( inorganic carbon ) is the only nutrient likely to be limiting for algal growth in lagoons. Substantial sludge accumulation in a lagoon may become soluble upon warming in the spring, releasing algal growth nutrients and causing an
algal bloom. Sludge resolution of nutrients is a major cause of high algal growth in a lagoon, requiring sludge removal
from the lagoon for correction.
The pH at a treatment lagoon is determined by the various chemical species of alkalinity that are present. The main
species present are carbon dioxide ( CO 2 , bicarbonate ion HCO 3 - , and carbonate
ion CO 3 - 2 ). High amounts of CO 2 yield a low lagoon pH, while high amounts of
CO 3 - 2 yield a high lagoon pH. Bacterial growth on BOD releases CO 2 which
subsequently dissolves in water to yield carbonic acid ( H 2 CO 3 ). This rapidly dissociates to bicarbonate ion, increasing the lagoon alkalinity. Bacterial oxidation of BOD causes a decrease in lagoon pH due to
CO 2 release.
Algal growth in lagoons has the opposite effect on lagoon pH, raising the pH due to algal use for growth of inorganic
carbon ( CO 2 and HCO 3 - ). Algal growth reduces the lagoon alkalinity which may
cause the pH to increase if the lagoon alkalinity ( pH buffer capacity ) is low. Algae can grow to such an extent in
lagoons ( a bloom ) that they consume for photosynthesis all of the CO 2 and HCO 3 - present, leaving only carbonate ( CO 3 - 2 ) as the pH buffering species. This causes the pH of
the lagoon to become alkaline. pH values of 9.5 or greater are common in lagoons during algal blooms, which can lead to lagoon effluent pH violations ( in most states this is pH = 9 ). It should be noted that an increase in the lagoon pH
caused by algal growth can be beneficial. Natural disinfection of pathogens is enhanced at higher pH. Phosphorus removal
by natural chemical precipitation is greatly enhanced at pH values greater than pH = 8.5. In addition, ammonia stripping
to the atmosphere is enhanced at higher pH values ( NH 3 is strippable, not NH 4 + ).
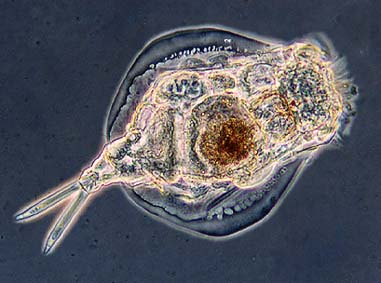
Protozoans and Microinvertebrates...
Many higher life forms ( animals ) develop in lagoons. These include protozoans and microinvertebrates such as rotifers, daphnia, annelids, chironomids ( midge larvae ), and mosquito larvae ( often termed the zooplankton). These organisms
playa role in waste purification by feeding on bacteria and algae and promoting flocculation and settling of particulate material. Protozoans are the most common higher life forms in lagoons with about 250 species identified in lagoons to
date (Curds, 1992). Rotifers and daphnia are particularly important in controlling algal overgrowth and these often
"bloom" when algal concentrations are high. These microinvertebrates are relatively slow growing and generally only
occur in systems with a detention time of > 10 days. Mosquitos grow in lagoons where shoreline vegetation is not removed
and these may cause a nuisance and public health problem. Culex tarsalis, the vector of Western Equine Encephalitis in
the western U.S., grows well in wastewater lagoons (USEPA, 1983). The requirement for a minimum lagoon bank slope and
removal of shoreline vegetation by most regulatory agencies is based on the public health need to reduce mosquito
vectors.
 "Lagoon Microorganisms"...
"Lagoon Microorganisms"...





 "Lagoon Microorganisms"...
"Lagoon Microorganisms"...

